9 Congenital Heart Disease
With few exceptions, animals with CHD have cardiac murmurs, and these are typically heard on auscultation, usually in a pup presented for routine evaluation and immunization. Of the few clinically consequential malformations that do not usually result in a heart murmur, most cause other clinical signs such as cyanosis, exercise intolerance, respiratory distress, or failure to thrive.
A continuous murmur heard over the left craniodorsal aspect of the heart base is the auscultatory hallmark of a PDA. The murmur is often but not always loud and is usually relatively coarse; it peaks in intensity at or about the second heart sound and then fades during diastole. This murmur is sometimes referred to as a machinery murmur. Continuous murmurs must be distinguished from to-and-fro or “bellows” murmurs, which consist of separate systolic and diastolic murmurs. In dogs, to-and-fro murmurs most commonly result from endocarditis of the aortic valve or ventricular septal defects that are complicated by aortic valve regurgitation.
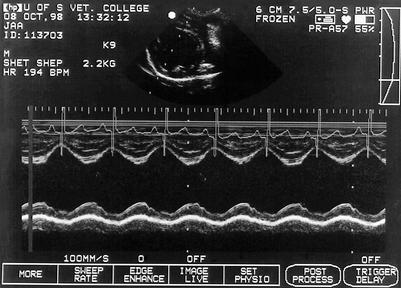
In animals with moderate or large PDAs (left-to-right shunts), dilation and hypertrophy of the left ventricle is evident echocardiographically (Fig. 9-1). Echocardiographic examination also demonstrates left atrial enlargement. Myocardial dysfunction, reflected in a large end-systolic ventricular dimension and often a low fractional shortening index, is sometimes evident. Direct visualization of the PDA is usually possible when the study is performed by an experienced echocardiographer. The PDA extends from the descending aorta to the bifurcation of the main pulmonary artery. It is best demonstrated by a cranial left parasternal image of the pulmonary artery bifurcation.
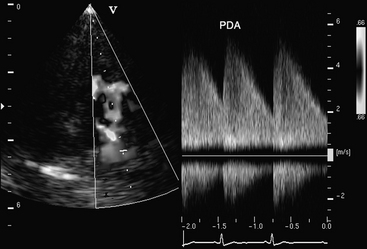
Doppler flow studies are used to confirm that blood from a PDA flows into the main pulmonary artery. In animals with a PDA, this will be evident on color-flow Doppler studies as a diastolic color mosaic that originates near the pulmonary artery bifurcation and extends retrograde toward the pulmonic valve. In many cases, the jet extends beyond the pulmonic valve and results in mild pulmonic regurgitation. On spectral Doppler studies, continuously disturbed flow will be evident within the pulmonary artery (Fig. 9-2).
PDA is essentially a physical diagnosis. When a continuous murmur is present in a dog with a typical signalment, the cause is almost always a PDA. However, Doppler echocardiographic examination is recommended as a noninvasive means of confirming the diagnosis. The echocardiographic evaluation of systolic function may also provide prognostic information that can influence therapy (see Fig. 9-3). Additionally, echocardiography provides a noninvasive means of detecting concurrent cardiac defects that might have an impact on prognosis and management.