CHAPTER 9 Clinical Examination of the Nonpregnant Equine Female Reproductive Tract
COMPONENTS OF THE EXAMINATION
External Evaluation
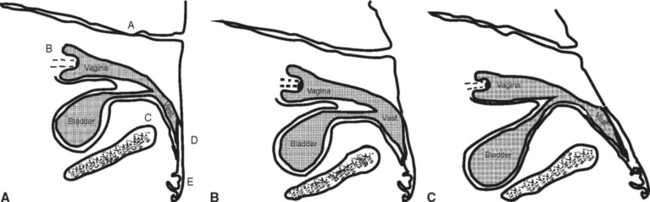
External evaluation of mares should begin with an examination of the mammae: their size, a comparison of the halves, and the gross appearance of the teats. Attempts should be made to determine whether their appearance can be explained by the age, parity, and lactational status of the mare. Abnormalities of the perineum, including integrity of the anal sphincter and vulvar conformation, can predispose mares to aspiration of air and feces into the genital tract.1 An assessment of vulvar conformation should include the relationship of the dorsal vulvar commissure to the pubis, the adequacy of the seal formed by the vulvar labia, and the angle of the vulva (Fig. 9-1). A windsucking test performed by gently parting the labia and listening for the sound of air rushing into the vagina can identify mares that would benefit from Caslick’s vulvoplasty.1 The examination must evaluate the ability of the three anatomic barriers (vulvar labia, vestibular sphincter, and cervix) to protect the uterus from the external environment and ascending infections. If a mare has foaled previously, an abnormality of the perineum may be evidence of trauma during parturition. Such mares are candidates for detailed vaginal examination to determine whether there is additional internal damage such as rectovaginal fistulae or lacerations of the vagina or cervix. The examiner should record the general appearance of the patient, taking note of body condition and conformation. If a mare has a very flat croup, she may have correspondingly poor vulvar conformation. The normal female phenotype may be altered by certain conditions such as X-chromosome monosomy, chronic use of anabolic steroids, and hermaphroditism.2–6 If the mare is a maiden, it is important to conduct a vaginal exploration prior to first cover.
Internal Evaluation
Additional means of identifying abnormalities in infertile mares are uterine cultures to determine the microbiologic status of the uterine lumen, endometrial cytologic examination to establish the presence of inflammatory cells, endometrial biopsy to detect histologic changes that could inhibit the mare’s ability to sustain a fetus to term, and uterine endoscopy to identify gross lesions within the uterine lumen that might prevent conception or interfere with pregnancy maintenance. The incidence of oviductal disease is very low in mares when compared with other domestic species, such as the cow.7 Although there have been some interesting reports that attach significance to collection of material within the uterine tubes or to inflammation of the uterine tubes, lesions of the oviducts do not appear to be common causes of reproductive failure in mares.8,9
Effect of Season
The reproductive activity of both mares and stallions is driven primarily by day length10–12 and to a much lesser extent by ambient temperature. In normal mares, seasonal and environmental changes are reflected in the findings during TRP. During winter anestrus the ovaries become inactive. The weight of ovarian stroma decreases and ovarian dimensions diminish.13 This normal change should not be mistaken for a congenital abnormality, but viewed within the context of the season. Ovarian development is a reflection of the activity, or lack thereof, of the hypothalamus and the pituitary gland. Ovarian activity (contrast inactive with the presence of follicles and corpora lutea) affects the balance of the genital tract, and in the absence of progesterone, uterine tone is poor (flaccid) and the cervix relaxed. With the coming of spring, day length increases, pineal gland suppression wanes, and activity of the hypothalamus and the anterior pituitary gland increases.10–12,14 With consistent photostimulation (increasing day length), the size and activity of the ovaries increase dramatically.13
During spring (vernal) transition, there is significant follicular growth, both in size and number.15 Transition occurs between winter anestrus and the ovulatory season (vernal transition) and again between the end of the ovulatory season and anestrus (autumnal transition).16
TRANSRECTAL PALPATION AND THE INTERRELATIONSHIPS OF SEASON, OVARIAN ACTIVITY, UTERINE TONE, AND CERVICAL RELAXATION
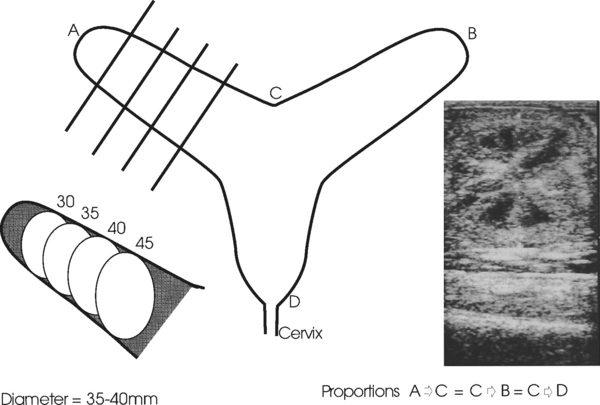
The nonpregnant uterus is T- or Y-shaped. The uterine horns approximate the cross-bar or the V of the letters. The uterine horns and ovaries are suspended by the broad ligaments (mesometrium and mesovarium) between the tuber coxae. The ovaries usually lie lateral and slightly ventral to the tips of the uterine horns. The ovaries are moveable within a range of 2 to 5 cm from the cranial border of the broad ligament and, by virtue of the mare’s motions or during palpation, may be brought to lie dorsal and medial to the broad ligament.
Ovarian Palpation
Structures that can routinely be palpated are follicles, ovulation depressions (OVD), corpora hemorrhagica (CH) for up to 5 to 6 days after ovulation (after which time they mature into functional corpora lutea and most are not palpable), and parovarian cysts.17–20 During winter anestrus the ovaries are small and inactive, there is an absence of follicles, and ultrasonographically the ovarian stroma appears uniform and completely unremarkable. With the onset of spring transition, follicular activity begins. The size and number of follicles can be highly variable during this period.21 Transition is associated with persistent follicles, which usually exhibit a slow rate of growth. Transitional follicles are anovulatory and their size and number can wax and wane.15,21 Unlike cystic ovarian disease in cows, which is a degenerative condition, persistent follicles in mares are normal structures associated with season. Transition ends with the first ovulation and an increase in serum progesterone.22,23 For the duration of the physiologic breeding season, mares experience an approximate 21-day cycle from one ovulation to the next.24,25 During the physiologic breeding season, follicles are produced in waves approximately every 10 days. Generally behavioral estrus is exhibited when serum progesterone concentrations fall below 1 ng/ml and is accompanied by a dominant follicle that increases in size at a rate of 5 to 6 mm per day until ovulation.14,26,27
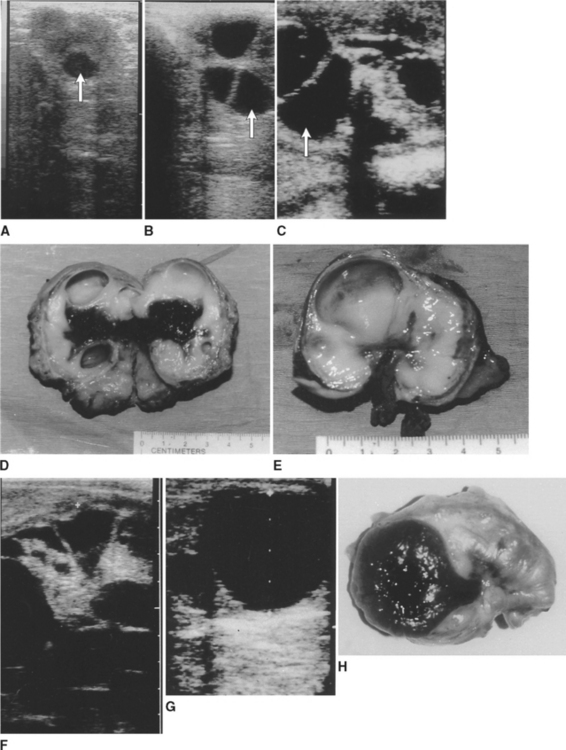
All ovulations in mares occur through the ovulation fossa; thus, developing follicles increase not only in width but also in depth as they extend through the ovarian stroma and encroach on the ovulation fossa (Fig. 9-2).28 Measurements of follicles recorded during an ultrasonographic examination may be larger if depth is measured rather than width. Thus, consistency in records will be achieved if measurements of dominant follicle size represent the width of the follicles, not their depth.27 Not all follicles soften prior to ovulation. As a follicle nears ovulation, it is possible to feel an irregular ridge around the perimeter of the ovulatory follicle where the ovarian stroma meets the edge of the follicle (Fig. 9-3, D).19 The ultrasonographic appearance of follicular fluid is uniformly black (see Fig. 9-2, G).29
Immediately after ovulation, the distinct cavity of an OVD can be palpated (see Fig. 9-3, E). Although not apparent in all mares, even in mares palpated at 6-hour intervals, when an OVD is detected, its roughened perimeter is palpable level with the ovarian surface, and its deepest point extends through the ovary to the ovulation fossa. In some instances, an OVD may be delineated as long as 18 hours, but in other cases blood fills the space (CH) much more quickly. It is possible to distinguish many follicles from CH during TRP by the angle formed between the edge of the follicle or the CH and the ovarian stroma. Except for some follicles that develop at the extreme poles of the ovaries, the palpable angle between a follicle and the ovarian stroma is approximately 90 to 100 degrees.19,27 In contrast, the angle between a CH and the stroma is much wider, 100 to 120 degrees (a CH is flatter than a follicle; see Fig. 9-3, D and F). CH also tend to be smaller than the follicle that preceded them.3 A CH feels softer and more fluctuant than the surrounding ovarian stroma during the first 1 to 2 days after ovulation. Ultrasonographically, the CH contains echogenic particles that result from blood filling the former follicular space. As the blood clots and organizes, its ultrasonographic appearance becomes more hyperechoic and, over the span of 2 to 4 days, more uniform.29 Although a mature CL cannot be palpated, it can consistently be distinguished by its uniform gray (more echogenic than the ovarian stroma) appearance during ultrasonographic examination (see Fig. 9-2, K).19,29
Parovarian cysts are embryologic remnants of the wolffian duct system.3 Their only significance is that they may be confused with follicles by the novice examiner. A careful transrectal examination can more specifically identify the location of these fluid-filled structures to be only “in the vicinity of ovaries,” that is, parovarian. The size of parovarian cysts increases either not at all or very slowly. They can range in size from a few millimeters to that of a modest-sized follicle.
Uterine Palpation
Excellent tone (ET): Upon palpation, the uterine horns are found to be distinctly tubular. Gentle digital indentation of the cranial ventral border of the uterine horns is met with resistance. The uterine horns feel similar to a rubber hose. ET would accurately describe the uterine tone expected in a heifer during estrus. ET in mares occurs first during early pregnancy (16–20 days of gestation) and occasionally during the first week postpartum (a reflection of optimal uterine involution). As pregnancy continues, the tone of the uterine wall immediately around the conceptus begins to decrease as the amount of fluid increases. ET may persist until 40 to 45 days from the middle to the tip of the nongravid horn.
Fair to good tone (FGT): Tubular, springs back, but more slowly.
Separate notations of tone are made for the left and right uterine horns if they differ by as much as a half grade, and separate values are recorded for diameters of the uterine horns if they differ by more than 5 mm. Individual mares may exhibit either good or fair tone during diestrus, and as they enter estrus, uterine tone begins to decrease. Typically, uterine tone decreases one grade by the time the mare ovulates. The uterus exhibiting good tone during diestrus will decrease to fair tone by late estrus. The uterus with fair tone during diestrus will decrease to poor tone near the time of ovulation. Following ovulation, uterine tone increases by one grade and returns to its former diestrual tone. In a decade of experience with 300 mares, individual mares during the physiologic breeding season tended to maintain similar “highs” and “lows” of uterine tone (GT ↔ FT, or FT ↔ PT) from one year to the next (C.L. Carleton, unpublished observations). As in any population there will be a few mares in which tone alterations between estrus and diestrus are less than one grade. Uterine horn diameter represents the average of approximately four sites between the tip (smallest diameter) and base (largest diameter) of each horn (Fig. 9-4). With practice this determination is quickly accomplished and becomes second nature for the examiner. This degree of detail can be especially useful in monitoring uterine involution in the first 8 to 30 days post partum.
Palpation of the Cervix
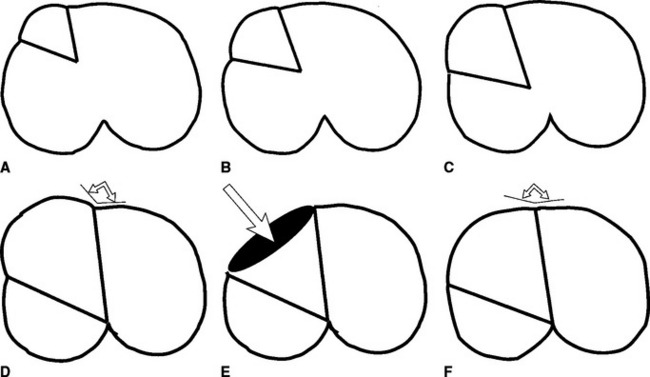
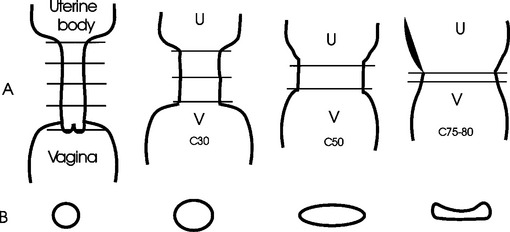
The dynamic capabilities of the cervix are quite extraordinary. Its proportions are influenced primarily by the presence (luteal phase) or absence (follicular phase) of progesterone. The cervix closes under the influence of progesterone.14,17 It relaxes, to a variable degree, during the estrous cycle when serum progesterone is low, by becoming shorter and wider from the beginning of estrus up until the time of ovulation, after which time it again closes. In contrast with the cow, in which the cervix is always palpable, the palpable characteristics of the equine cervix can vary from tightly closed and easily identifiable to not palpable, if it is completely (100%) relaxed.17,18 Consistent and accurate palpation of the cervix is the most difficult part to master of TRP of the genital tract, but persistence is worth while. To maximize accuracy of the description, cervical palpation is best left until the end of a detailed genital examination. If the palpator has adequately described the ovarian findings and defined the diameter and approximate length of the uterine horns, then the location of the internal cervical os can readily be anticipated. The length of a uterine horn (tip to bifurcation) is approximately equal to the length of the uterine body (the distance from the bifurcation of the uterine horns to the internal cervical os) (see Fig. 9-4). As the examiner’s hand is being withdrawn near the completion of a TRP, the caudal extent of the uterine body can be identified. By gently pinning of the uterine body under the outspread fingers and palm, the internal os of the cervix can be identified. There is a subtle inward coning at the transition of the uterine body to the internal os of the cervix (see Fig. 9-4). Except when the cervix is completely relaxed, the external cervical os can be identified transrectally. Using relative proportions of length to width, the closed cervix is described as having a ratio of 4:1. In the author’s records, cervical relaxation is routinely recorded in increments of 10% (C.L. Carleton, unpublished observations). Full definition of the cervix requires assessment of three factors: length, width, and tubularity (Fig. 9-5). This combination provides a quick and reasonable means of assessing the degree of cervical relaxation. The percent of relaxation of the cervix is related to the structures on the ovaries. The cervix and the uterus thus are additional predictors of the mare’s readiness for mating and ovulation. Most normal mares achieve at least 40% relaxation of the cervix prior to ovulation (C.L. Carleton, unpublished observations).
Prediction of the Time of Ovulation
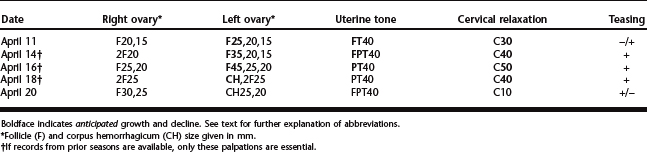
With serial palpations, an appreciation of the relationship of a dominant follicle to decreasing uterine tone and increasing cervical relaxation can improve the breeding management of mares. Knowledge of follicular size at ovulation in individual mares from records of previous seasons and estrous periods makes it feasible to predict more closely the time of ovulation. Insemination is avoided until the follicle is within 5 to 10 mm of its anticipated ovulatory size, When coupled with evidence of decreasing uterine tone and a relaxing cervix during estrus (Table 9-1), accurate, consistent, multiple-year records are invaluable in predicting the optimal time to breed and, during subsequent ovulatory seasons, result in a decrease in the number of covers per mare. Individual mares tend to ovulate similar-sized follicles year after year. The exception occurs during foal heat, during which the dominant follicle may become significantly larger. Nevertheless, the effect of the follicle on uterine tone and on cervical relaxation is consistent near the time of ovulation. Factors that prevent the cervix from responding to ovarian activity include anabolic steroid administration, induration of the cervix subsequent to chronic pyometra, and chronic cervicitis.4,5
Good records permit assessment of trends and recognition of abnormalities of a mare’s estrous cycle. Additional structures noted during TRP and ultrasonography may include endometrial folds (see Fig. 9-4, inset), uterine fluid, and uterine cysts. During estrus, the endometrial folds are particularly prominent owing to edema.17 Notations made of the location and size of lymphatic and glandular cysts will prevent confusion with pregnancy during an early ultrasonographic examination. If fluid is detected during TRP, its nature can be further defined by ultrasonography, microbiology, cytology, lavage, or endoscopy. An early report dealt with grading of luminal fluid and based its severity on its echogenicity.30 The volume of uterine fluid can be estimated ultrasonographically. Few mares show evidence of gross vaginal discharge even when a large volume of fluid is in the uterus.3 Once the presence of fluid has been confirmed, a uterine swab should be collected prior to performing other techniques because bacterial contaminants introduced during subsequent procedures could mask the original problem. Intraluminal adhesions, unless extraordinarily large or extensive, are not palpable.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree