Chapter 178 Basic Husbandry and Medicine of Pet Reptiles
Boas, pythons, king snakes, rat snakes, and milk snakes are the most common snakes kept as pets. Iguanas, geckoes, monitors, and bearded dragons are frequently encountered pet lizards. Amphisbaenians are small, worm-like reptiles not commonly found in the pet trade. Snakes, lizards, and amphisbaenians are all in the order Squamata. The order Chelonia has approximately 235 species and consists of turtles, tortoises, and terrapins. Of the chelonians, box turtles, red-eared sliders, and a variety of tortoises are kept most commonly as pets. The order Crocodilia has only 21 species, including alligators, crocodiles, caimans, and gharials.
comparative anatomy and physiology
Body Temperature
Table 178-1 PREFERRED OPTIMUM TEMPERATURE BASED ON HABITAT
| Habitat | Temperature Range (°F) |
|---|---|
| Tropical | 85–100 |
| Desert | 80–102 |
| Lowland temperate | 70–90 |
| Mountain temperate | 70–85 |
Table 178-2 PREFERRED OPTIMUM TEMPERATURE FOR COMMON REPTILES
| Common Species | Temperature Range (°F) |
|---|---|
| Boas and pythons | 75–95 |
| Rat/milk/king/garter snakes | 68–95 |
| Desert tortoises | 65–105 |
| Painted turtles | 55–95 |
| Box turtles | 65–90 |
| Anoles | 70–90 |
| Chameleons | 55–75 |
| Green iguanas | 75–105 |
Gastrointestinal Tract
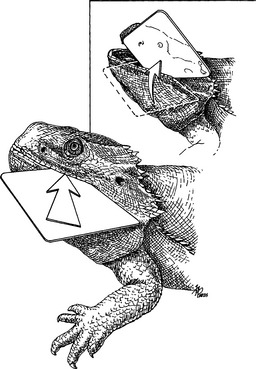
Never force open a reptile’s mouth. To open the mouth of a snake or lizard, place a plastic credit card or hard rubber spatula at the corner or front of the mouth and use gentle, prolonged pressure combined with a slight wiggling motion. Once the mouth is opened, slide the card or spatula across the jaws (Fig. 178-1). The beak of larger chelonians can bite through many plastics, so a thin, but sturdy metal spatula may be required to open the mouth. Be careful not to crack the beak. Anesthesia may be required to open the mouths of very large reptiles.
Respiratory System
Most reptiles lack a diaphragm, and breathing is accomplished by expansion of the chest wall by intercostal muscles (except turtles), pectoral limb movement (except snakes), and smooth muscle contraction in the lung. Crocodilians also use movement of the liver to create negative pressure. Chelonians use movement of front limbs and visceral organs instead of excursion of the chest wall, which is impossible because of their rigid shell. A minimal amount of oxygen can be extracted from water through the skin in certain species of all reptilian orders. Some aquatic turtles can exchange dissolved oxygen across their pharynx and cloaca while submerged, although these sources only provide maintenance levels of oxygen.
Cardiovascular
Renal System
Not all reptiles have a urinary bladder. The bladder is most highly developed in terrestrial chelonians and some lizards. The bladder acts as a reservoir to store urine and conserve water. Isosthenuric urine from the kidneys is diverted from the cloaca to the bladder, where water is reabsorbed. The concentrated urine then is routed back to the cloaca for excretion. In reptiles without a bladder, diluted urine is moved from the cloaca into the large colon for water absorption. Salts also may be reabsorbed.
Sex Determination in Common Pet Reptiles
Snakes
Lizards
In general, males become more brightly colored during the courtship season. Males have more highly developed dorsal spines, dewlaps, and horns. Male iguanas produce keratinaceous plugs along the insides of their thighs from glands called femoral pores. Their function is unknown, but they are thought to be used for marking territories and to stimulate females by rubbing (scratching) the femoral pores over the female’s hind quarters. Females have smaller versions of these pores. Only males of some gecko species have preanal pores that appear as little black pores located near the vent.
Immune System
Endocrine System
Special Sense Organs
HUSBANDRY
Caging
Aquatic
Never use an under-gravel filter because these may cause anaerobic toxins to accumulate in the gravel. Disturbance of the gravel or an overturn of tank water allows release of these toxins and may kill tank inhabitants. An out-of-tank biologic filter works best. Flush the sand, gravel, or fibers in these systems frequently to remove gross debris. Even these systems often cannot handle both fecal and discarded food loads. Therefore, offer food for limited periods in a separate tank, then return the reptile to its usual enclosure. The feeding tank does not need a filter because the water is changed completely before and after each feeding session and/or group of animals. Disinfect the tank and allow it to dry after feeding to prevent waste buildup and transmission of disease.
Nutrition
Carnivore
Herbivore/Omnivore
RESTRAINT
Lizards and Crocodilians
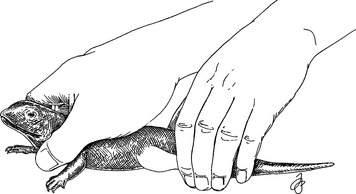
Restrain small lizards and crocodilians by using the thumb and forefinger to grasp the caudal mandibular area. Cradle the dorsal body in the palm of the same hand. Control the abdomen, chest, and pelvis with the remaining fingers, thus allowing for respiration (Fig. 178-2).
Grasp medium-sized lizards and crocodilians over the dorsal surface of the head and neck with one hand, and grab the base of the tail and pelvis with the other hand (Fig. 178-3). Wrap the reptile securely in a towel to prevent the back legs from scratching. If necessary, restrain the back legs by taping them to the tail. To capture mildly aggressive animals, use a towel as a blind to hide the restrainer’s hand from view. If an aggressive animal is brought in a sack, use the sack as a blind and grasp the head through the sack. Peel the sack away from the body without losing control of the head. A snare or snake hook can be used to pin the head if the animal is extremely aggressive. Care must be taken to use just enough pressure to prevent escape but not enough to cause trauma. Large lizards and crocodilians can inflict serious bites and often use their powerful tails as whips. Tape the snout of aggressive crocodilians shut with duct tape. The masseter muscles are extremely strong; the digastricus muscle, which opens the jaw, is very weak.
Snakes
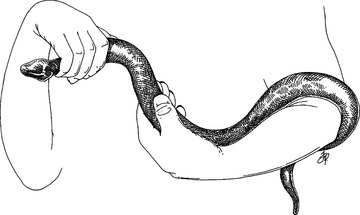
Most non-aggressive snakes may be picked up mid-body and allowed to glide from hand to hand. Some snakes are more secure if allowed to coil around an arm (Fig. 178-4). Most snakes struggle less when handled with minimal restraint. Never let any snake near your face or around your neck!
Chelonians
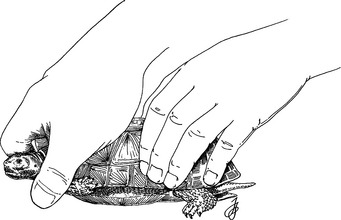
Hold chelonians by the dorsal caudal portion of the shell (Fig. 178-5). This placement makes it difficult for the animal to reach around and bite, or to scratch with its rear feet. Be sure to have a firm grip, especially if the turtle is wet. Always hold a chelonian over a table. Dropping a turtle may cause severe life-threatening shell fractures. Handle soft-shelled turtles with rinsed examination gloves, thus preventing damage to the delicate shell. Snapping turtles and some other long-necked turtles can reach the rear of their shells with their beaks. Grasp these turtles by the base of the tail. Hold them with tail in one hand and the front of the carapace just over the head in the other hand. Large snapping turtles are very dangerous to handle and should be left to experts.