Chapter 173 Avian Reproductive Tract Disorders
Avian reproductive disorders are a result of complex combinations of hormonal, physiologic, and behavioral actions reacting to photoperiods, food availability, and availability of nest sites. Environmental influences unique to captivity may induce reproductive and hormonal activity in several ways. For instance, artificial lighting may interfere with the normal photoperiod and annual light cycles, resulting in inappropriate cycling. Food is typically available ad libitum in captivity, and it often is high-fat, calorically dense seed or foods high in simple carbohydrates, such as corn and fruit. These foods may actually stimulate reproduction. A lack of an appropriate mate may also cause reproductive problems. Most pet birds are not intended for breeding and do not have mates. As a result, some of these birds select an abnormal mate such as their human cohabitants or cage furniture. Along with all of these common environmental influences, there may be genetic factors that contribute to a lack of normal reproductive hormonal balance.
FEMALE REPRODUCTIVE DISORDERS
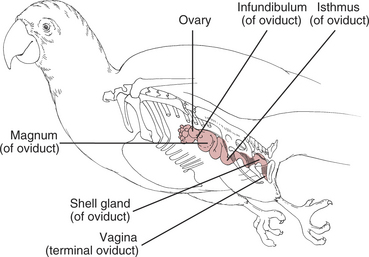
See Figure 173-1 for reproductive anatomy of the female bird.
Chronic or Excessive Egg Laying
Etiology
Diagnosis
Diagnosis of chronic egg laying is based on history and physical examination.
Physical Examination
Treatment
Medical therapy for chronic egg laying focuses on stopping egg production while removing predisposing stimuli and correcting any secondary diseases that may be present. Pharmacologic, behavioral, nutritional, environmental, and surgical options are used alone or in combination, depending on the needs of the individual patient. Pharmacologic options have included medroxyprogesterone acetate, levonorgestrel, human chorionic gonadotropin, testosterone, and leuprolide acetate.
Pharmacologic Therapy (Table 173-1)
Leuprolide Acetate
Table 173-1 MEDICAL THERAPY FOR REPRODUCTIVE DISORDERS
| Drug | Dosage | Comments |
|---|---|---|
| Leuprolide acetate | 150–800 mg/kg IM | Administered every 14 days; three doses are usually adequate |
| Human chorionic gonadotropin | 250–500 IU/kg IM on days 1, 3, and 7 | Stable in refrigerator 60 days |
| 500–1000 IU/kg IM | If a second egg is laid, repeat dose on day 3; if a third egg is laid, repeat dose on day 7 | |
| Levonorgestrel | Not recommended | |
| Medroxyprogesterone | Not recommended | |
| Arginine vasotocin | 0.01–1.0 mg/kg IM | Stable in standard freezer |
| Prostaglandin E (Prepidil Gel) | 0.1 ml/100 g | May freeze into aliquots and thaw |
| 0.002–0.1 mg/kg | Applied topically prior to administration; relaxes uterovaginal sphincter while inducing uterine contractions | |
| Prostaglandin F-2-alpha (Lutalyse) | 0.02–0.1 mg/kg IM | May not relax uterovaginal sphincter when inducing uterine contractions |
| Topically applied to prolapsed uterine tissue to stop hemorrhage and shrink tissues |
Human Chorionic Gonadotropin
Other Hormonal Treatments
Environmental Modification
Polyostotic Hyperostosis
Etiology
Diagnosis
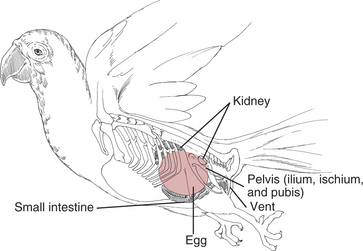
Egg Binding and Dystocia
Definition
Etiology
Clinical Signs
Diagnosis
Physical Examination
Diagnostic Imaging
Laboratory Evaluation
Treatment
Therapy varies with history, severity of clinical signs, and diagnostic test results.
Supportive Care
Pharmacologic Therapy (See Table 173-1)
Prostaglandin E-2
Prostaglandin F-2-alpha and Other Hormonal Therapy
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree