Chapter 10 Avian Influenza
DEFINITION AND TYPES
Avian influenza A virus (AIV) is a member of the Orthomyxoviridae family and may be classified according to the antigenicity of its surface proteins hemagglutinin (H) and neuraminidase (N), and on the basis of its pathogenicity in chickens after intravenous inoculation. Currently, 16 H (H1-H16) and 9 N (N1-N9) subtypes have been described in avian species.19 Individual subtypes may be composed of any combination of one of the H and one of the N proteins.
Highly pathogenic avian influenza (HPAI, formerly “fowl plague”), an acute, generalized disease in which mortality may be as high as 100%, is restricted to subtypes H5 and H7, although not all viruses of these subtypes necessarily cause HPAI. All other AIV strains are low-pathogenic avian influenza (LPAI) virus strains and cause a much milder, primarily respiratory disease with loss of egg production.7 In certain cases the LPAI virus phenotype (of subtype H5 or H7) may mutate into the HPAI virus phenotype by the introduction of basic amino acid residues (arginine or lysine) at the cleavage site of the precursor hemagglutinin (HA0),3 which facilitates systemic virus replication. H5 and H7 subtypes with an amino acid sequence at the HA0 cleavage site comparable to those that have been observed in virulent avian influenza viruses are considered HPAI viruses, even when mortality in chickens is low. However, the two forms of avian influenza (HPAI and LPAI) are distinctly different and should be regarded as such.
ETIOLOGY AND EPIDEMIOLOGY
Avian influenza virus has a worldwide distribution and is infective for all avian species (commercial, domestic, and wild), with variable morbidity per virus isolate and species. Aquatic avian species, mainly those of the taxonomic orders Anseriformes (ducks, geese, and swans) and Charadriiformes (shorebirds, gulls, and terns), are considered the main natural reservoir of all avian influenza viruses, including the LPAI ancestral viruses of HPAI strains.32 Replication of LPAI viruses occurs mainly in the intestinal tract, with excretion of high virus loads, and efficient transmission via the fecal-oral route.55 Experimentally infected mallards have been shown to excrete virus asymptomatically for up to 17 days.59 It has been shown that AIV remains infectious in feces for up to 32 days17 and in lake water for 4 days at 22°C, to more than 30 days at 0°C.55 Migrating waterfowl are thought to carry LPAI viruses over long distances and may initiate outbreaks of HPAI by the introduction of these LPAI viruses into poultry flocks, which subsequently change into HPAI viruses.56 Recent HPAI H5N1 viruses have been predominantly associated with oropharyngeal shedding4,45; the impact of this on environmental contamination, persistence, and transmission is still unknown.
Poultry species (e.g., chickens, turkeys, quail, ostriches) are generally highly susceptible to infection with HPAI virus. In 2002 an outbreak of HPAI H5N1 virus occurred in wild migratory avian species and resident waterfowl, and the high pathogenicity in ducks was confirmed in laboratory infections.44 Since 2002, this particular HPAI virus subtype has made an unprecedented spread from Southeast Asia throughout Asia and into Europe and Africa, with morbidity and mortality not only in domestic poultry, but also in a large number of avian species, specifically, 96 species (spp.) from 14 orders: Anseriformes (31 spp.), Charadriiformes (5), Ciconiiformes (6), Columbiformes (3), Falconiformes (9), Galliformes (9), Gruiformes (4), Passeriformes (18), Pelecaniformes (2), Phoenicopteriformes (1), Strigiformes (4), Struthioniformes (1), Psittaciformes (1), and Podicipediformes (2 spp.).52 Outbreaks along the recognized flyways from Southeast Asia into Europe have suggested that this HPAI virus subtype may be distributed directly by migrating waterfowl, and HPAI virus infections have been detected in several migratory species.10,30,62 However, it remains clear that domestic waterfowl,9,24,45 specific farming practices, agro-ecologic environments, and transportation of domestic avian species or their products with trade at local markets have played key roles in the amplification and spread of HPAI H5N1 virus in Asia.21,42
Several mammalian species (ferrets, horses, pigs,9 seals, and humans13,18) had been reported with infections with the H5 and H7 subtypes of AIV up to 1997. The recent HPAI H5N1 virus subtype has caused mortality in (experimentally) infected Owston’s banded palm civets (Chrotogale owstoni),39 domestic dogs,43 domestic cats (Felis domestica),29,38 leopards (Panthera pardus) and tigers (Panthera tigris),28,50 stone martens (Martes foina), and ferrets (Mustela putorius furo)52 and has caused 256 human cases with 151 deaths to date (October 2006).61
Noteworthy are the fatal HPAI H5N1 virus infections with severe pneumonia of domestic cats, tigers, and leopards that fed on infected poultry,28 because felids had previously been considered resistant to disease after AIV infection.23 Horizontal spread of infection was suspected50 and has been demonstrated experimentally in domestic cats,29 with excretion from both the respiratory and the intestinal tract.38
Fatal infections of domestic dogs with an H3N8 strain usually found in horses have been documented recently, and this virus is apparently spreading and has potential to become endemic in dogs.12
CLINICAL SIGNS
Low-pathogenic AI infections of wild birds usually produce no clinical signs. In poultry, LPAI is characterized by mild to severe respiratory signs, excessive lacrimation, decreased egg production, and signs of generalized malaise (ruffled feathers, depression, decreased water and feed intake). Disease caused by HPAI virus infection may vary depending on species, age, strain of virus, and environmental factors. Infections of poultry by HPAI viruses are characterized by a drop in egg production, inappetence, depression, respiratory signs, sinusitis, watery diarrhea, excessive lacrimation, edema of comb and wattles with cyanosis and hemorrhages, neurologic signs, and high mortality. Other avian species may have a higher resistance to infection, with no clinical signs shown, death soon after showing signs,53 or slow recovery until no clinical signs are seen.4
AIV infection in mammalian species is predominantly an upper respiratory tract infection with some lung involvement. Signs of infection include sneezing, nasal discharge, malaise, and pyrexia. HPAI H5N1 virus–infected wild felids and laboratory cats have shown additional signs of respiratory distress, serosanguineous nasal discharge, protrusion of the third eyelid, conjunctivitis, neurologic signs, and death.28,29,38,50
Postmortem Lesions
Gross lesions are variable, depending on host species, pathogenicity of the virus, and presence of secondary pathogens. Whereas lesions caused by LPAI in poultry are mostly found in the respiratory tract (especially the sinuses) and coelomic cavity (egg-yolk peritonitis), lesions caused by HPAI are more diverse. If death is peracute, no gross lesions may be observed. In poultry, subcutaneous edema of the head, upper neck, and feet may be accompanied by hemorrhages and cyanosis, particularly in the wattles and combs. Hemorrhages may also be found in visceral organs, especially the epicardium, pectoral muscles, and the mucosa of the proventriculus and ventriculus. Necrotic foci are most common in the pancreas, spleen, and heart. Lungs have focal ventral to diffuse interstitial pneumonia with edema and may be congested or hemorrhagic.46
Viral-induced lesions in HPAI H5N1 virus–infected ducks and gulls were only found in those with clinical signs. Lesions found (petechial hemorrhages in pancreas, ventriculus, apex of heart, and cerebrum) were milder in birds that recovered.4
Histologic lesions consist of multiorgan necrosis and inflammation (mainly brain, pancreas, heart, lung, and primary and secondary lymphoid organs).4,11,46 The most common lesions in recovered birds are lymphoplasmacytic perivascular encephalitis and heterophilic pancreatitis.4
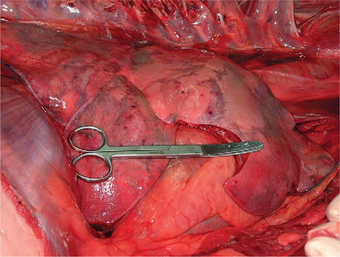
Surprisingly, experimental H5N1 virus infection in cats was demonstrated to be systemic and not limited to the respiratory system. Gross lesions consisted of congestion of the lungs and enlargement of, with multifocal petechial hemorrhages in, the tonsils, mandibular lymph nodes, retropharyngeal lymph nodes, and liver.38 Infected tigers grossly had severe congestion of the lungs with hemorrhages, serosanguineous exudates throughout tracheal and bronchiolar lumina, and pleural effusion. Histologic lesions consisted of severe diffuse lung hemorrhages and edema, nonsuppurative meningoencephalitis, and moderate multifocal necrotizing hepatitis28,29,38,50 (Figure 10-1). Extrarespiratory spread in laboratory cats included tissues of the nervous, cardiovascular, urinary, digestive, lymphoid, and endocrine systems and was associated with severe pathologic changes, consisting of necrosis and inflammation.38
DIAGNOSIS
Material for Laboratory Analysis
Cloacal and oropharyngeal swabs may be taken to show presence of virus or viral RNA in live birds. Small birds may be harmed by swabbing, and for these species, collection of fresh feces is an adequate alternative. Cotton swabs should be placed in vials with virus transport medium60 (e.g., Hank’s balanced salt solution with 10% vol/vol glycerol, 200 U/mL penicillin, 200 μg/mL streptomycin, 100 U/mL polymyxin B sulfate, and 250 μg/mL gentamicin, at a final pH of 7.0-7.4) and kept on wet ice. Samples may be stored at 4°C for up to 4 days; prolonged storage should be at −80°C.
Suspensions in antibiotic solutions of organ material, swabs, or fecal material may be inoculated in embryonated chicken eggs, which are incubated and tested for hemagglutinating activity. The conventional isolation in chicken eggs and characterization is still the method of choice for the World Organization for Animal Health (OIE),34 but molecular techniques for the detection of viral RNA by RT-PCR techniques have been developed, which are much faster, generating reliable subtype-specific results within a few hours, and may provide complementary (copy) deoxyribonucleic acid (cDNA) for nucleotide sequencing.34
A human rapid enzyme immunoassay (EIA) kit (Directigen Flu-A, Becton-Dickinson Microbiological Systems, Alphen aan den Rijn, Netherlands), which uses a monoclonal antibody to detect the nucleoprotein of influenza A strains, has been reported to detect viral antigen in avian specimens as well.40 This test takes 20 to 30 minutes and therefore might be useful for screening. However, the test lacks sensitivity, has not been validated for different avian species, and subtype identification is not achieved.
Serum may be collected for the detection of antibodies to nucleocapsid or matrix antigens by means of agar gel immunodiffusion tests. These antigens are antigenically similar in all influenza A viruses, but not all birds develop detectable titers.34 Hemagglutination inhibition (HI) tests, which are subtype specific, may be used for routine diagnostic testing, for epidemiologic studies, and to determine vaccine efficacy.
MANAGEMENT, THERAPY, AND PREVENTION
Highly pathogenic avian influenza (HPAI) viruses, but also H5 and H7 LPAI, are categorized as notifiable diseases by the OIE, based on their potential for rapid spread, serious economic or public health consequences, and impact on the international trade of animals and animal products. Most countries have banned vaccination of domestic poultry because of interference with eradication policies and international trade agreements, but they will allow regulated vaccination in emergencies. Eradication measures during an outbreak in domestic poultry include (long-term) confinement, stamping out of all poultry on the infected farm, preemptive culling of animals on neighboring farms, and emergency vaccinations (European Union [EU] Directive 92/40/EEC).
Surveillance of wild birds may provide early warning signs for the introduction of HPAI virus.33 Several countries have initiated surveillance campaigns of free-ranging wild birds. Wild bird populations that experience high mortality rates should be submitted to national or regional reference laboratories for testing (for a European listing, see the EAZWV Handbook of Infectious Diseases20). Birds showing clinical signs may be captured, isolated, and selectively culled when testing is positive for HPAI virus. There is no scientific basis for large-scale culling of free-ranging wild bird populations to control outbreaks or their spread, and it would be highly undesirable from a conservationist perspective.36 Instead, measures should be taken to prevent poultry from coming into contact with wild birds.
In zoos a balance should be found between what control measures are effective, practical and realistic, and socially tolerable. The standard measures used in poultry would be detrimental to the welfare and breeding programs of valuable and endangered avian species in zoos. Increased biosecurity is the first line of defense during outbreaks of HPAI and may be complemented by vaccination. Accreditation of zoos (e.g., by AZA, EAZA, or other national/international organizations) has resulted in standardized high levels of biosecurity, decreasing the risk of introduction and increasing the likelihood of containment of infectious diseases. With an outbreak of HPAI, however, levels of biosecurity should always be raised immediately, with hygienic measures implemented accordingly, to prevent entrance or spread of the virus. Attention should focus on both exclusion (identification and elimination of possible routes of entrance, e.g., by live birds, cages, equipment, clothing) and containment (reduction of the risk of infection for neighboring cages) of the virus. Detailed guidelines have been written by the Association of Zoos and Aquariums (AZA)1 and the Wildlife Conservation Society (WCS).58
Disinfection
Other means of virus inactivation include the following2,17:
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree