49 AVIAN HEMATOLOGY
2 What are some important principles to understand about the identification of avian leukocytes by blood smear evaluation?
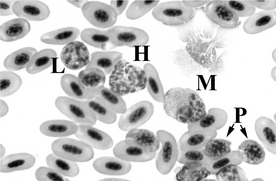
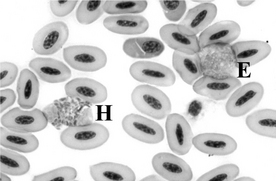
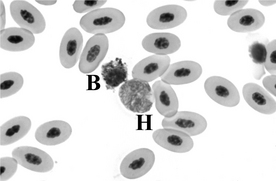
3 Describe the various avian leukocytes.
5 What are common methods of enumerating total numbers of leukocytes?
For faster, automated, direct quantification, automated cell counters are available based on flow cytometric technology. These automated counters are capable of quantifying the total number of leukocytes in avian blood. However, known drawbacks of this technology include the potential for including nucleated erythrocytes and thrombocytes in the leukocyte count and, generally, inconsistent automated differential cell counts. Manual examination of a blood smear is therefore essential when performing differential cell counts in conjunction with an automated cell counter. Flow cytometric technology is expensive and available primarily to commercial laboratories with the time, finances, and dedicated personnel to utilize and service the technology appropriately. As with any automated technology daily quality control/quality assurance measures must be in place to produce reproducible, accurate results. (For detailed discussion on leukocyte count techniques, see the Bibliography following Chapter 53.)
6 What causes heterophilia?
In general, heterophilia may be caused by the following three conditions, acting singly or in combination (Box 49-1):
Box 49-1 Causes of Heterophilia in Birds
Chronic myelogenous leukemia is a rarely seen cause of neutrophilia in domestic animals, and to my knowledge, it is undescribed in pet birds.
11 What are the expected complete blood count (CBC) findings in birds with heterophilia of physiologic leukocytosis?
13 What are the expected CBC findings in birds with heterophilia of inflammatory disease?
Mild heterophilia may be the only abnormality in mild inflammatory disease. With increasing severity, left shift (increased presence of immature heterophils, e.g., band forms, metamyelocytes, myelocytes, promyelocytes), heterophil toxicity (cytoplasmic basophilia, abnormal vacuolation, abnormally shaped cytoplasmic granules, degranulation, large heterophils), and greater numbers of heterophils may be present and reflect a significant demand for heterophils in tissues. Monocytosis may also be observed. In severe inflammatory disease (e.g., chlamydophilosis, mycobacteriosis), total leukocyte counts may reach or exceed 100,000 cells/μl. Keep in mind that overwhelming inflammation may result in heteropenia with left shift and heterophil toxicity (see following questions). The numbers of lymphocytes may be decreased, normal, or increased with inflammatory diseases.
14 What are the predominant causes of heteropenia?
Heteropenia may be caused by the following three conditions, acting singly or in combination (Box 49-2):
16 What causes decreased bone marrow heterophil production?
Heterophil production abnormalities may be the result of destruction of heterophil precursors, hormonal/chemical suppression of granulopoiesis, or myelophthisic processes. Destruction of heterophil precursors in birds may be caused by viral infection or adverse drug reactions (e.g., fenbendazole in painted storks, piperacillin and/or doxycycline in one budgerigar). Suppression of granulopoiesis may occur with hormonal treatment (progesterone) and other myelosuppressive therapy (cyclophosphamide, cancer chemotherapies, radiation). Myelophthisic processes, such as neoplastic disease (leukemia, lymphosarcoma), may infiltrate the bone marrow and displace hematopoietic precursors.
19 What causes lymphocytosis?
Lymphocytosis may be caused by the following three conditions, acting singly or in combination (Box 49-3):
20 How do physiologic leukocytosis, antigenic stipulation, and lymphoproliferative disease result in lymphocytosis?
24 What are causes of lymphopenia?
Lymphopenia is primarily caused by the following three conditions, acting singly or in combination (Box 49-4):
25 How do corticosteroids, inhibition of lymphocyte production, and acute infection result in lymphopenia?
26 Describe the mechanisms of lymphocyte redistribution and lymphocyte trapping in lymphoid tissues.
Corticosteroids (endogenous or exogenous) promote redistribution of lymphocytes from the CP to the MP and other locations (bone marrow, lymphoid tissues). Under the influence of inflammatory cytokines released during acute inflammation, and sometimes in combination with endogenous corticosteroid release, lymphocytes may be redistributed from the CP to the MP or other locations (bone marrow, site of inflammation, lymphoid tissue). Inflammatory cytokines may also promote the temporary sequestration of lymphocytes in lymphoid tissues as part of the antigen presentation process.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree