Model
Method
Sample/preparation
References
Impact acceleration
Gelatin zymography
Cortex
Frozen/lysis buffer
Nonidet P-40
Ding et al. 2009 (1)
Clinical population
Severe TBI
Gelatin zymography
Fluorometric assay
Plasma/CSF
CSF protein extracts
Lysis buffer/Tris, SDS
Grossetete et al. 2009 (2)
Intracerebral hemorrhage
Gelatin zymography
Frozen/cortex
Lysis buffer/TritonX-100
Gelatin sepharose 4B
Affinity purification
Grossetete and Rosenberg 2009 (3)
Closed head injury
Weight drop
Gelatin zymography
Cortical punch
Lysis buffer Brij 35
Triton X-100
Gelatin sepharose 4B
Affinity purification
Homsi et al. 2009 (4)
Hypoxia/ischemia
Cortical development
Gelatin zymography
Cortical hemispheres
Frozen/lysis buffer
Nonidet P-40
Ranasinghe et al. 2009 (5)
Peripheral thermal injury
Gelatin zymography
Whole brain
Total protein extracts
Reyes et al. 2009 (6)
Clinical population
Moderate/severe TBI
Gelatin zymography
Plasma
Frozen/lysis buffer
Nonidet P-40
Vilalta et al. 2008 (7)
Spinal cord injury
Gelatin zymography
In situ zymography
Spinal cord
Frozen/Brij 35 lysis buffer
Gelatin sepharose 4B
Affinity purification
Yu et al. 2008 (8)
Closed head injury
Weight drop
Juvenile rat
Gelatin zymography
Cingulate, parietal cortex
Thalamus
Frozen/lysis buffer
Brij 35, Triton X-100
Gelatin sepharose 4B
Affinity purification
Sifringer et al. 2007 (9)
Blade lesion of cortex
Gelatin zymography
Frontal/parietal cortex
Brainstem, cerebellum
Total protein extracts
Yhamaguchi et al. 2007 (10)
Entorhinal lesion
Fluid percussion injury
Fluorometric assay
Hippocampus
Lysis buffer/Tris
Kim et al. 2005 (11)
Fluid percussion injury
Hypothermia
Gelatin zymography
In situ zymography
Frozen/cortex, hippocampus, thalamus
Lysis buffer, Tris/SDS
Truettner et al. 2005 (12)
Subarachnoid hemorrhage
In situ zymography
Striatum
Sheba et al. 2004 (13)
Controlled cortical impact
Gelatin zymography
Whole brain/cortex
Fresh frozen/lysis buffer
Lysis buffer/Nonidet P-40, SDS
Mori et al. 2002 (14)
Scratch in vitro injury
Gelatin zymography
Culture media supernatant
Lysis buffer/SDS
Wang et al. 2002 (15)
Entorhinal lesion
Fluid percussion injury
Gelatin zymography
Hippocampus
Lysis buffer/T-PER
Phillips and Reeves 2001 (16)
Aspiration corticectomy
Gelatin zymography
Frozen/perilesional cortex
Lysis buffer/TritonX-100
Vecil et al. 2000 (17)
Controlled cortical impact
Casein zymography
Cortex
Lysis buffer/cytosolic extracts
Zhao et al. 1998 (18)
While several excellent overviews of these zymographic methods already exist for general reference (3, 20), we have found that direct application of these methods to tissue samples generated by traumatic brain injury (TBI) models may be problematic. In working with these methods, we have modified the standard gelatin zymography protocol in order to optimize assessment of MMP activity following brain injury. This chapter focuses on these modifications, which we have used to more confidently track gelatinase activity following TBI. We have not yet applied reverse zymography or in situ zymography to our models; however, others have done so successfully to reveal details of ECM response after TBI, subarachnoid hemorrhage, and spinal cord injury (8, 12, 13). The reader is referred to these publications for details as to how this additional methodology may be adapted for brain injury analysis. In the present chapter, the major procedural steps that we have applied to produce reliable results with gelatin zymography are described first. These include (1) sample preparation, (2) gel electrophoresis and incubation, (3) visualization of lysis, (4) measurement of signal, and (5) considerations of experimental design. Additionally, we discuss several aspects of zymographic analysis in brain injury models which deserve consideration in advance of experimentation. Foremost, depending upon model and level of injury, there may be challenges in pulling out detectable signal from standard tissue extracts. Moreover, different brain regions may also vary considerably in the extent or pattern of MMP signal. As described below, we have varied extraction procedures and extended the lysis development phase of the protocol to aid in signal detection. In our studies, at least two additional benefits for zymographic analysis have been observed relative to TBI. First, we have used the method to assay ECM-related mechanisms which might be part of specific therapeutic manipulations. Our data supports clear differences in MMP activity as a function of specific postinjury drug treatment. Second, the separation of pro and active enzyme species may reveal interesting spatio-temporal changes during injury–recovery cycles. These data can be combined with protein and RNA profiles to significantly enhance the extent of interpretation.
2 Materials
2.1 Sample Preparation
All extraction and zymography buffers are prepared just prior to use. All reagents are of ACS grade.
2.1.1 Tissue Dissection
1.
Standard dissection tools, including razor blades for blocking, forceps, microtip probes, and fine tip iridectomy scissors
2.
Platform for dissection chilled over ice
3.
Sterile Ringer’s solution
2.1.2 Hippocampal Tissue Extraction
1.
Pellet pestles with microtubes and pellet pestle cordless motor (Kimble Chase Life Science and Research Products, Vineland, NJ, #749520-0090)
2.
Tissue Protein Extraction Reagent Thermo Scientific (T-PER) (Thermo Scientific, Rockford, IL, #78510)
3.
Pierce BCA Protein Assay Kit (Thermo Scientific, #23225)
2.1.3 Extraction of Corpus Callosum and Cortex
RIPA Lysis Buffer (Millipore, Billerica, MA, #20-188)
2.2 Gel Electrophoresis and Incubation
2.2.1 Gelatin Gels
1.
Novex 10% Zymogram Gelatin Gels (10-well, 0.1% gelatin, Tris–Glycine, Invitrogen, Carlsbad, CA, #EC6175)
2.
Tris–Glycine SDS Running Buffer (10×) (Invitrogen, #LC2675)
3.
Tris–Glycine SDS Sample Buffer (2×) (Invitrogen, #LC2676)
2.2.2 Gel Incubation
1.
Zymography Renaturing Buffer (Invitrogen, #LC2670)
2.
Zymography Developing Buffer (10×) (Invitrogen, #LC2671)
2.3 Visualization of Lysis
1.
Brilliant Blue R (Sigma-Aldrich, #B7920-50G)
2.
Destain solution 1: Methanol:glacial acetic acid:nanopure water (4:1:5, v:v:v)
3.
Destain solution 2: Methanol:glacial acetic acid:nanopure water (0.7:1:8.3, v:v:v)
2.4 Measurement of Signal
1.
G:Box ChemiHR System (SynGene)
2.
GeneSnap Software (SynGene)
2.5 Experimental Design
2.5.1 Standards and Inhibitors
1.
MMP-2 Standard (Millipore, Billerica, MA, #CC071); proenzyme = 68 kDa nonreduced and active (59–62 kDa)
2.
Purified human MMP-9 (Millipore, #CC079); proenzyme = 88 kDa nonreduced and mature active form = 82 kDa
3.
N-ethylmaleimide (NEM) (Sigma-Aldrich, St.Louis, MO, #E3876)
4.
Phenylmethyl sulfonyl fluoride (PMSF) (Sigma-Aldrich, #P7626)
2.5.2 Activation of Pro-MMPs
p-aminophenylmercuric acetate (APMA; Sigma-Aldrich, #A-9563)
2.5.3 Drug Manipulations
1.
MK-801 (Sigma-Aldrich, #M-107)
2.
Minocycline Hydrochloride (Sigma-Aldrich, #286710)
3 Methods Description
3.1 Sample Preparation
3.1.1 Tissue Dissection
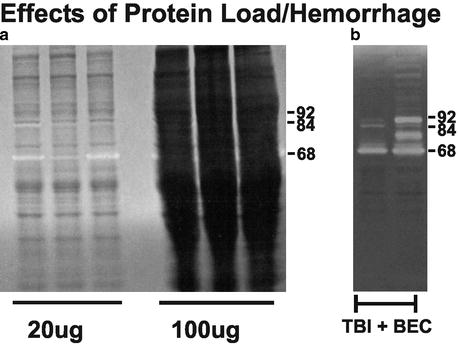
Under general anesthesia (4% isoflurane in carrier 70% N2O, 30% O2), animals are decapitated, the brain rapidly removed, and regions of interest dissected on an iced platform (Note 1). Depending upon desired sample site, the brain may be blocked first to gain better access for dissection. Rinse the dissected tissue with sterile saline before extraction to remove any excess surface blood (Fig. 1; Notes 2 and 3). Fresh tissue is then homogenized directly in extraction buffer (for our samples, T-PER, RIPA, or Brij). We routinely dissect hippocampus, cerebral cortex, and corpus callosum from cases subjected to unilateral entorhinal cortical lesion (UEC), moderate central fluid percussion TBI, or combined TBI and bilateral UEC (TBI + BEC), as well as paired sham-injured animals. This method permits successful extraction of tissue gelatinases using a variety of buffer systems, both with and without detergent (Notes 4 and 5; see also Table 1. for details of published extraction buffers).
 < div class='tao-gold-member'>
< div class='tao-gold-member'>





Only gold members can continue reading. Log In or Register to continue
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


